CTI Invents Thermochromic Inks Free of Bisphenol A, F and S in Response to Pharmaceutical Demand
Press release from the issuing company
Colorado Springs, Colo. – As the world’s supply chain for pharmaceutical drugs comes under increasing scrutiny for safety and quality, Chromatic Technologies Inc. (CTI) has introduced printable thermochromic technology that is free of Bisphenol A, F and S. The development meets the stringent regulatory standards for pharma packaging.
The spread of the coronavirus has refocused the world’s attention on proper care of the supply chain for vaccines and drugs. Additionally, interest has surged in low-cost technology that monitors the cold chain, identifies temperature abuse and tampering and provides authentication.
Bisphenol A (BPA) has been labeled “an endocrine disruptor,” though it is found in many plastics, the lining of food and beverage cans and carbonless paper. Several companies have attempted to limit Bisphenol A, F and S in order to be more cautious. CTI recognized that effort and has responded with its latest innovation now available to pharmaceutical packaging companies.
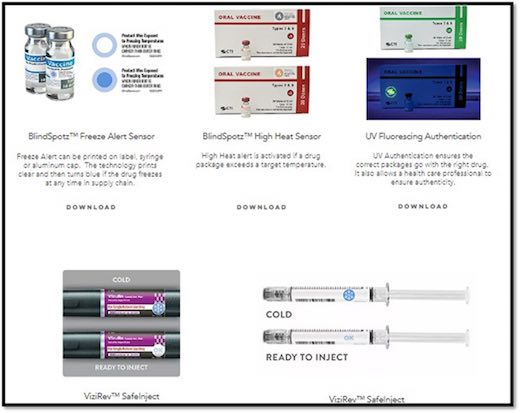
CTI’s BlindSpotz™ technology is a portfolio of patented technology that created low-cost printable sensors for drugs that detects freezing, thawing, gradual warming, tampering and authentication.
Pharmaceutical printers, as well as food and beverage printers, are requiring CTI’s innovative thermochromic inks to be free of Bisphenol A, F and S. Heretofore, printers have rejected traditional thermochromics for not meeting strict internal and external regulatory standards.
“We undertook the challenge of reinventing thermochromic technology to meet the high standards of pharmaceutical customers,” said Lyle Small, CTI’s chief executive officer. “Not only did we create CTI’s products free of BPA, F and S, but we also installed a 21-step checklist to validate quality and performance.” For full compliance information, visit www.ctiinks.com.

WhatTheyThink is the official show daily media partner of drupa 2024. More info about drupa programs
© 2024 WhatTheyThink. All Rights Reserved.









Discussion
Join the discussion Sign In or Become a Member, doing so is simple and free