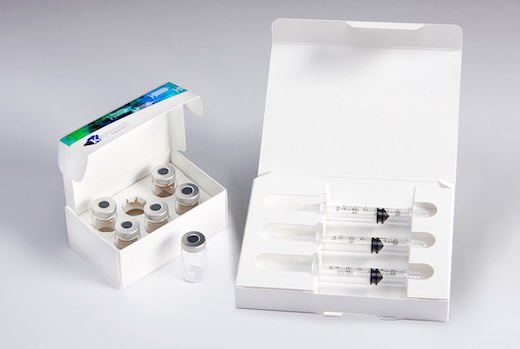
Family of secondary packaging options ideal for prefilled syringes, autoinjector pens, vials & ampoules.
Newark, NJ – Keystone Folding Box Co., a designer and manufacturer of paperboard packaging solutions, now offers a line of secondary packaging systems for injectable pharma products. Available in a variety of tailored features and in both all-paperboard and plastic-hybrid formats, solutions are available for prefilled syringes, autoinjector pens, vials and ampoules, as well as combinations of these.
Biologics, biosimilars, vaccines and other medications delivered through injection present unique secondary packaging challenges. Addressing these complexities, Keystone’s collection meets a range of needs through options such as tamper evidence and child-resistance via reclosable locking mechanisms – all while ensuring product protection throughout the supply chain. The portfolio includes:
The Vial Pack line of cartons includes designs for packaging a single vial or ampoule, or as many products as a program requires. The easy-to-store packs allow ample real estate for detailed usage instructions – a must for healthcare professionals and caregivers, including staff in hospitals, assisted living facilities and at-home care settings.
A line of Injectable Packs is suitable for prefilled syringes or autoinjector pens, and can be designed to include ancillary items like sterile wipes.
The InjectaSlide package incorporates a thermoformed tray that slides inside a carton and into a locked position; a lock release button must be pressed to unlock and slide the tray forward for easy access to each dose.
Keystone’s secondary packaging solutions for injectables are appropriate for automated or manual production, and are ideal for everything from clinical trials and academic settings to mail order drugs, compounding pharmacies and other full-scale commercialization scenarios. The packaging is suitable for use in temperatures as low as -80C which is typical with cold-chain distribution required of injectable products.
Child-resistant features are available with several of the solutions and, for outpatient or in-home use, multi-dose packs can be designed capable of prompting patients to administer the correct dose at the proper date and time.
“Medications administered by injection are often high-value and delivered in fragile primary packaging such as vials or prefilled syringes. Injectable products require protection that recognizes and respects these circumstances, and that can accommodate the various shapes, sizes and special features our packaging solutions provide,” said Ward Smith, Director of Marketing & Business Development at Keystone Folding Box Co. “Our diverse, customizable range of secondary packaging solutions for injectable products exemplifies Keystone’s commitment to ensuring product protection and enhanced consumer experiences in every design.”
To learn more about Keystone Folding Box Company, please contact Ward Smith at Keystone Folding Box Company, at (513) 871-4747, [email protected] or visit www.keyboxco.com.



